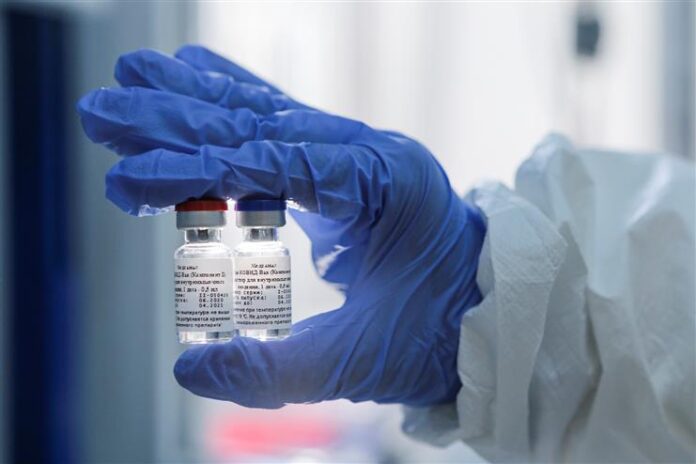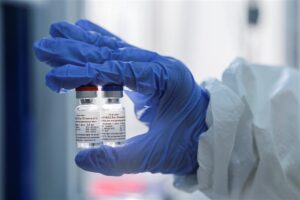
The Russia Direct Investment Fund (RDIF), which is steering Russia’s Sputnik V competitor immunization, right now in Phase 3 preliminaries, has collaborated with the Hyderabad-based Dr. Reddy’s Laboratories to test, and subject to administrative endorsements in India, flexibly 100 million dosages of the antibody.
G.V. Prasad, co-executive and overseeing chief, Dr. Reddy’s Laboratories, said in a joint explanation with RDIF: “We are satisfied to cooperate with RDIF to carry the immunization to India. Phase 1 and 2 outcomes have indicated guarantee, and we will lead Phase 3 preliminaries in India to meet the prerequisites of the Indian controllers. Sputnik V antibody could give a believable choice in our battle against COVID-19 in India.”
The RDIF said in the announcement, “The Sputnik V antibody, which depends on very much examined human adenoviral vector stage with demonstrated security, is going through clinical preliminaries for the COVID pandemic. Conveyances might start in late 2020 subject to consummation of fruitful preliminaries and enlistment of the antibody by administrative experts in India.”
The understanding does exclude producing the immunization in India. An RDIF representative said the association was all the while searching for makers.
Dr. Reddy’s-RDIF understanding comprises the second significant worldwide arrangement for providing an expected antibody after the arrangement between British organization AstraZeneca and the Pune-based Serum Institute of India (SII). The last will fabricate 100 million dosages of the antibody, of which purportedly half will be for India.
On August 11, the Sputnik V antibody, created by the Gamaleya National Research Institute of Epidemiology and Microbiology, was enrolled by the Ministry of Health of Russia and turned into the world’s originally enlisted immunization against COVID-19 dependent on the human adenoviral vectors stage. Nitty-gritty data on the immunization, the mechanical foundation of human adenoviral vectors, and different subtleties are accessible at sputnikvaccine.com.